Buy GMP Documents SOP and Manuals
Document listing:
All listed documents have unique document numbers and descriptions. Documents are listed in the shopping cart according to their categories. Some categories have multiple sub-categories. Individual documents are listed directly under these categories or sub-categories, as shown in the table.
The document number starts with a prefix (e.g., VAL for Validation) matching the category followed by a sequential number. Most documents have sample links to download for your review.
Click the “Buy Now” button below.
Shopping cart:
To buy the documents, select the number & description by navigating through the categories and sub-categories.
Add the documents to your shopping cart. Similar to standard online shopping carts, you may add, remove, update, and checkout.
Purchased documents are instantly downloadable and editable Word files (.docx). The links are sent to your email.

Quality Assurance and Compliance SOPs
In this category you will find SOPs for establishing quality management and compliance systems such as management and control of master documents, reporting of deviations and non-conformance, quality concern investigation process, product complaint handling procedure, quality audit, vendor assessment, evaluation and certification, rework, return and rejects, procedures, GMP training, quality risk management, product review and many other to meet your need.

Manufacturing, Packaging Operation SOPs
This category have a number of SOPs on production operations including gowning requirements for production zones, cleaning responsibilities and methods for employees, pest control, production logbooks, tag out procedure, line clearance, line cleaning, yield and reconciliation of components and finished goods, cleaning of vessels and IBCs, tablet packing machine construction & operation, manufacturing instruction for tablet packing and many more.

Quality Control Laboratory SOPs
This category lists procedures on retest dating of raw materials, calibration of laboratory instruments, laboratory documentation, management of reference substances, GLP requirements of laboratory workbook, managing analytical reagents & retention samples, waste management, laboratory supplier approval, laboratory results-out of specification investigation, raw materials finished goods-laboratory testing methodology etc.
Microbiology Laboratory SOPs
List of SOPs on entry procedure to sterile filling areas, validation of aseptic gowning, microbiological data recording, destruction of biological waste, depyrogenation of glassware, media preparation, aseptic media filling and micro. integrity leak (soup) testing procedure and guideline, environmental and plant hygiene monitoring procedure, microbial limit testing etc.
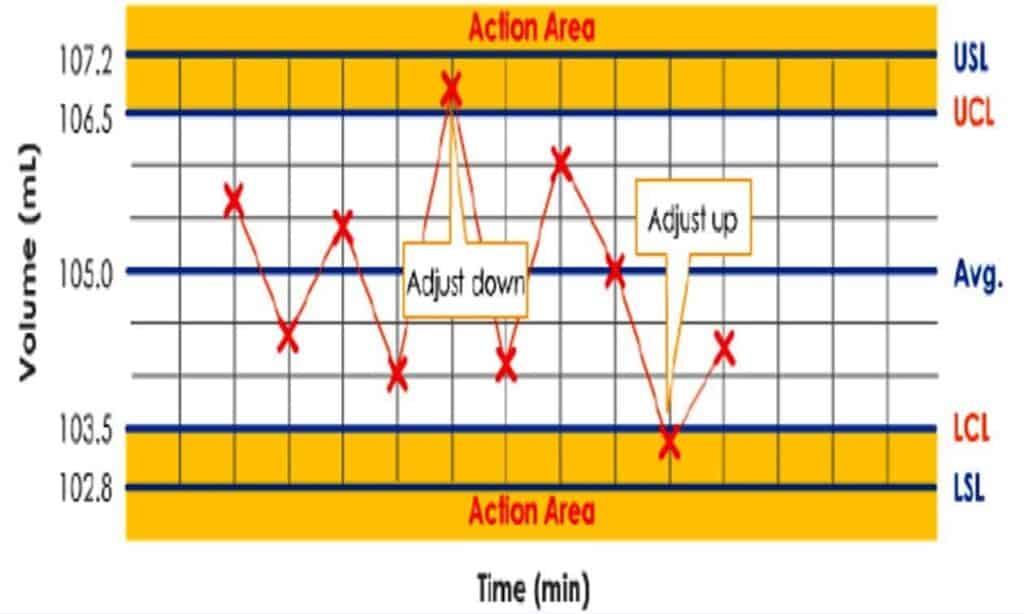
Process, Cleaning, Method & Computer Validation SOPs
This category has a list of procedures on validation systems including validation-concept and procedure, re-validation, process validation guideline, validation master plan, test method validation, cleaning validation, validation of laboratory instruments, equipment qualification, computerized systems validation, impact assessment, design qualification, facility and utility validation etc.

Warehouse Management SOPs
Warehouse SOP list that includes receipt of incoming goods, raw material and components-incoming-handling by sampler, procedure for warehouse to processing issues, returns and rejects, dispatch of goods, inventory management, warehouse locations and storage area, finished goods transfer to quarantine and distribution warehouse, sampling of raw materials, components and printed materials, work in progress area, safety procedure of warehouse racking, forklift operation in warehouse, dispensing procedure etc.

Manuals - Analytical & Microbiology Laboratory
Best industry practice and guidance documents on chemistry and microbiology laboratory management that covers from environmental monitoring, sterility testing, preparation & maintenance of stability protocols, stability master plans, commercial stability testing of APIs, explanation of repeat testing and retesting, reference & retention samples, analytical procedures and validation and many others.

Manuals - Quality Management Systems
Best industry practice and guidance documents on product quality complaint handling, application of quality risk management, statistical rationale for raw material sampling, quality agreements, structured on-the-job training system, disposal of rejected and waste materials, compliance improvements plans, warehousing and distribution of commercial products, reduced testing program and many more.
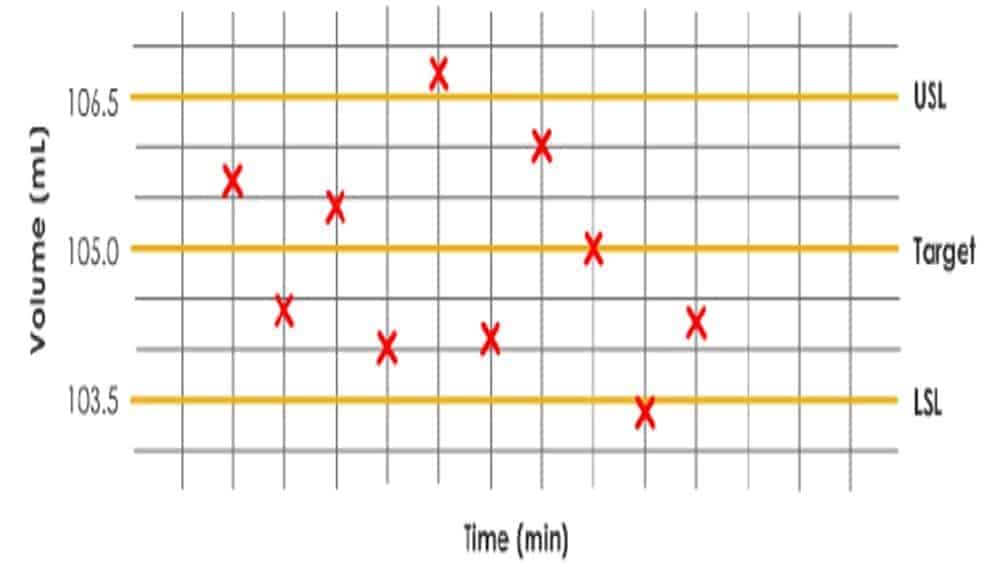
Manuals - Process, Cleaning, Method & Computer Validation
Best industry practice and guidance documents on preparation of validation master plan, clean equipment hold times establishment and practices, process validation of bulk drug and formulated products, documentation models for continuous quality verification, matrices and bracketing of medicinal products in process validation, performance qualification versus process validation, the validation of facilities and system and plenty more.

Manuals - Sterile, Non Sterile Manufacturing Operations
Best industry practice and guidance documents on management of non-sterile active pharmaceutical ingredient, manufacturing documentation, maintenance and calibration of GMP critical item, gamma radiation sterilization, retreatment and blending of API & formulated product, preventing cross contamination, returned goods, receipt handling and storage of starting & packaging materials and a lot more.

Manuals - GMP Auditor Training
List of GMP training manuals on auditing principles for GMP audit, personnel & training system, deviation management system, validation system, change management system, complaint system, documentation system, calibration, preventative maintenance & housekeeping system, computerized systems, utilities system, warehouse and distribution system, environmental monitoring system, microbiology and sterility testing laboratory, analytical quality & stability testing laboratory, material handling system, active API manufacturer, packaging material vendors, packaging and labeling operation, aseptic sterile area, etc.

GMP Quality & Validation Templates
List of shelf ready commonly used templates on quality and validation.

GMP Quality Management Software
Shelf ready GMP compliant access database applications on deviation management, product complaint management, change control management, out of specification result investigation and non-conforming product management.
